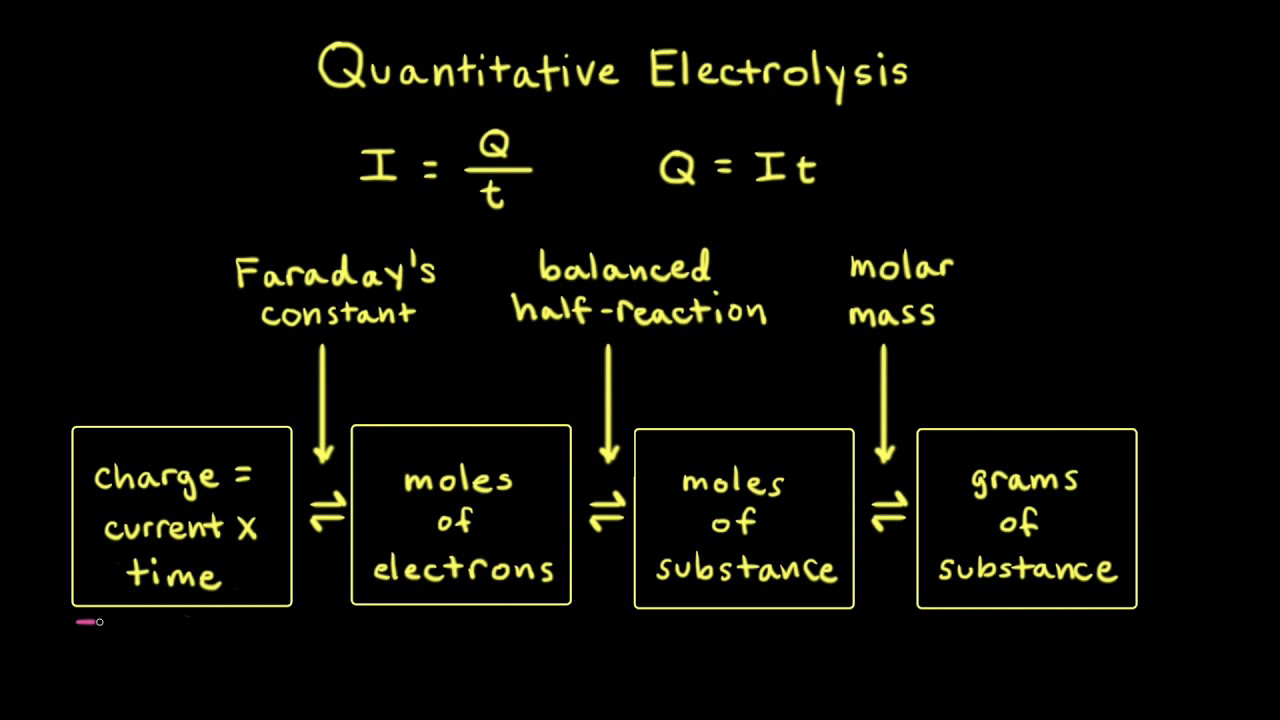
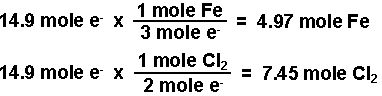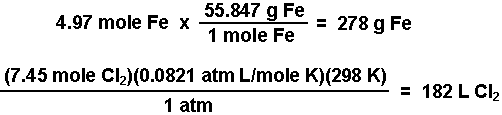SOLVED: Electrolysis of aqueous solution of 0.1 M CuBrz was carried out using inert electrode at 25 Determine the products of electrolysis at anode and cathode: Justify. Calculate the mass of product
Welcome to Chem Zipper.com......: During electro refining of Cu how much time is needed to produce 250g Cu on the cathode if the current is kept at 11 A?