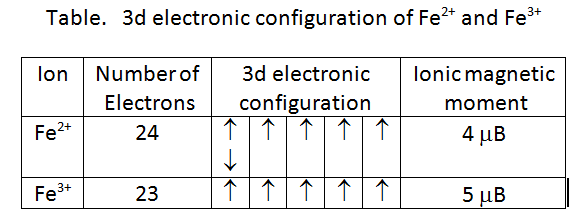
Which of the following ion has maximum magnetic moment ? 1.Mn^3+ 2.Cu^2+ 3. Fe^3+ 4.v^3+ Also explain what is magnetic moment ?
Amongst the following ions which one has the highest magnetic moment value? (i) [Cr(H2O)6]3+ (ii) [Fe(H2O)6]2+ - Sarthaks eConnect | Largest Online Education Community
What will be the theoretical value of spin only magnetic field when Fe(SCN)3 reacts with the solution containing F ions to yield a complex

What is magnetic moment and how can we find magnetic moment of Fe3+!! Pls someone help me !! - Chemistry - - 15334915 | Meritnation.com

Illustration of the net magnetic moment calculation in various types of... | Download Scientific Diagram
![The spin-only magnetic moment of `[Fe(NH_(3))_(6)]^(3+) and [FeF_(6)]^(3-)` (in units of BM ) - YouTube The spin-only magnetic moment of `[Fe(NH_(3))_(6)]^(3+) and [FeF_(6)]^(3-)` (in units of BM ) - YouTube](https://i.ytimg.com/vi/KVj56QvOV1I/maxresdefault.jpg)
The spin-only magnetic moment of `[Fe(NH_(3))_(6)]^(3+) and [FeF_(6)]^(3-)` (in units of BM ) - YouTube
![Explain why [Fe(H(2)O)(6)]^(3+) has magnetic moment value of 5.92 BM where as [Fe(CN)(6)]^(3-) has a value of only 1.74 BM ? Explain why [Fe(H(2)O)(6)]^(3+) has magnetic moment value of 5.92 BM where as [Fe(CN)(6)]^(3-) has a value of only 1.74 BM ?](https://d10lpgp6xz60nq.cloudfront.net/ss/web/584381.jpg)
Explain why [Fe(H(2)O)(6)]^(3+) has magnetic moment value of 5.92 BM where as [Fe(CN)(6)]^(3-) has a value of only 1.74 BM ?






![6)]^(3+)` has magnetic moment value of 5.92 BM whereas `[Fe(CN) 6)]^(3+)` has magnetic moment value of 5.92 BM whereas `[Fe(CN)](https://i.ytimg.com/vi/h9yxC3M4DSM/maxresdefault.jpg?sqp=-oaymwEmCIAKENAF8quKqQMa8AEB-AH-CYAC0AWKAgwIABABGGUgVChVMA8=&rs=AOn4CLAIfOZpXbuvpM77ZlcUR5tTbbLZLA)




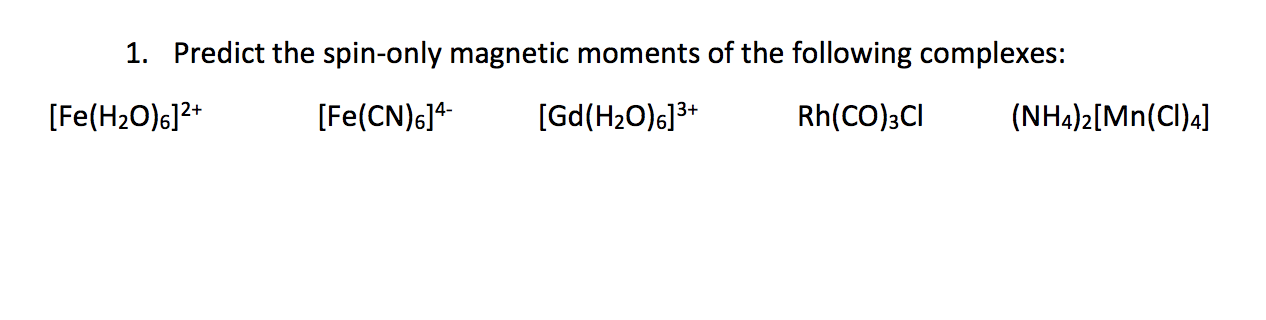

![The spin magnetic moment of iron in K3 [ Fe (CN)6 ] is : The spin magnetic moment of iron in K3 [ Fe (CN)6 ] is :](https://haygot.s3.amazonaws.com/questions/1952807_1117559_ans_880cbb7d24bb4d01ab49a890bc90ed14.jpg)