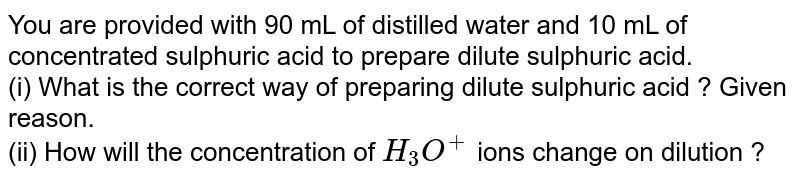You are provided with 90 mL of distilled water and 10 mL of concentrated sulphuric acid to prepare dilute sulphuric acid. What is the correct way of preparing dilute sulphuric acid? Give reason.

SOLVED: What volume of 18 M sulfuric acid must be diluted with distilled water to prepare 500.0 mL of0.50 M H2SO4? A) 0.014 mL B) 0.14 mL 14 mL 9 14 mL E) 140 mL