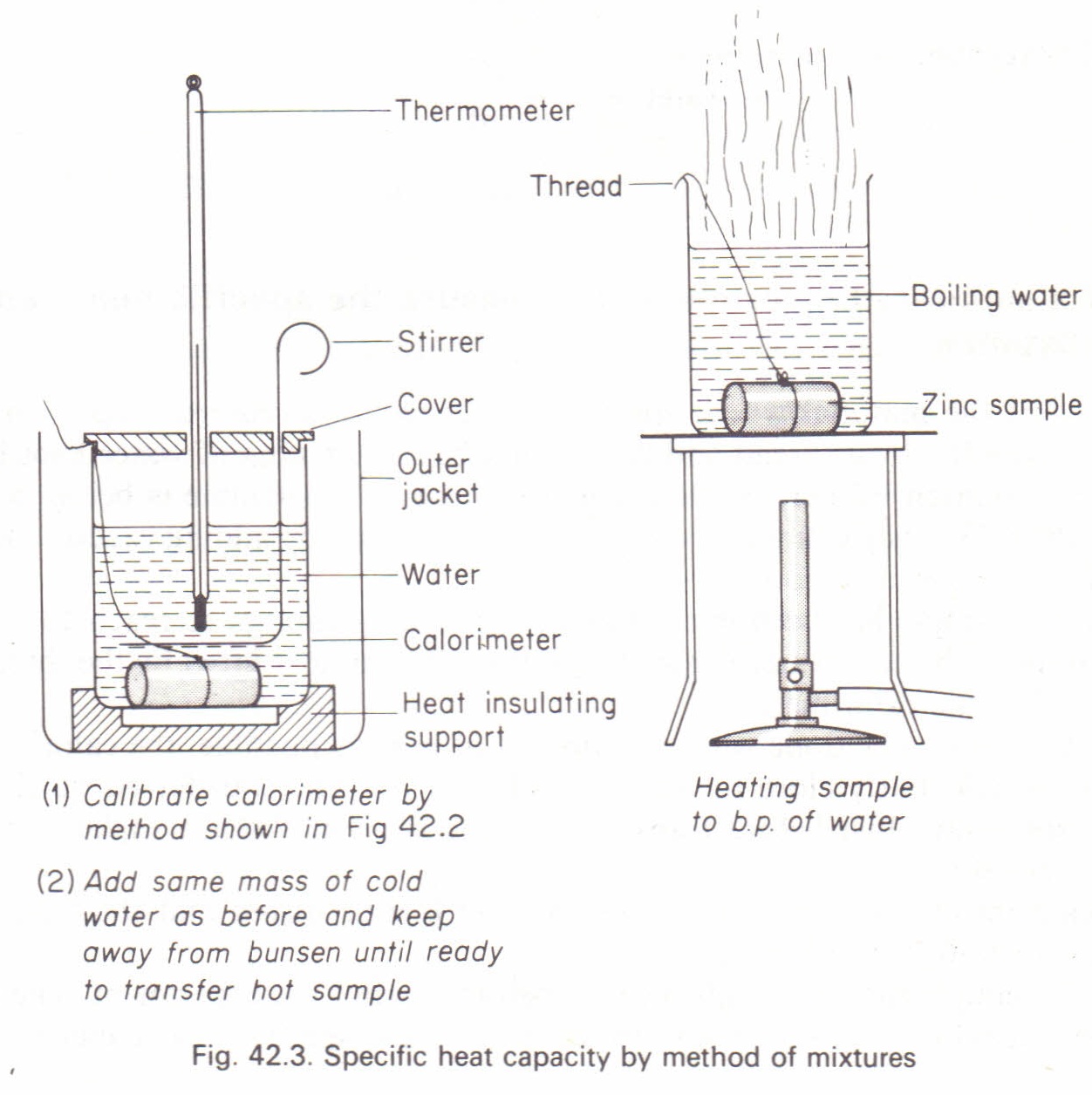
CHAPTER 4 HEAT EFFECT. Consider the process of manufacturing ETHYLENE GLYCOL (an antifreeze agent) from ethylene : -Vaporization -Heating Ethylene (liquid) - ppt download

Thermal fluids with high specific heat capacity through reversible Diels-Alder reactions - ScienceDirect

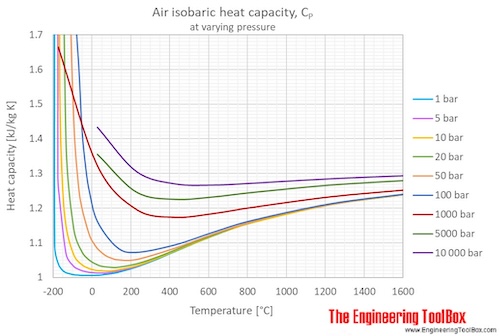
Percentage Deviations of Isobaric Heat Capacities Calculated for Dry... | Download Scientific Diagram

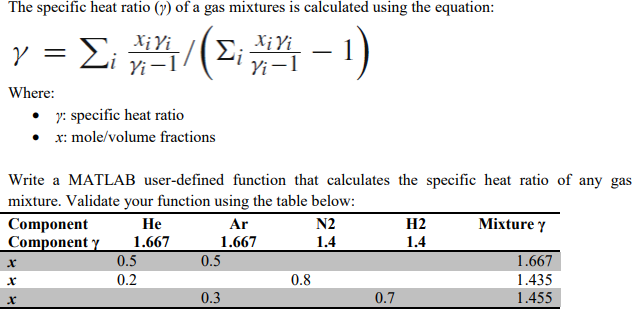
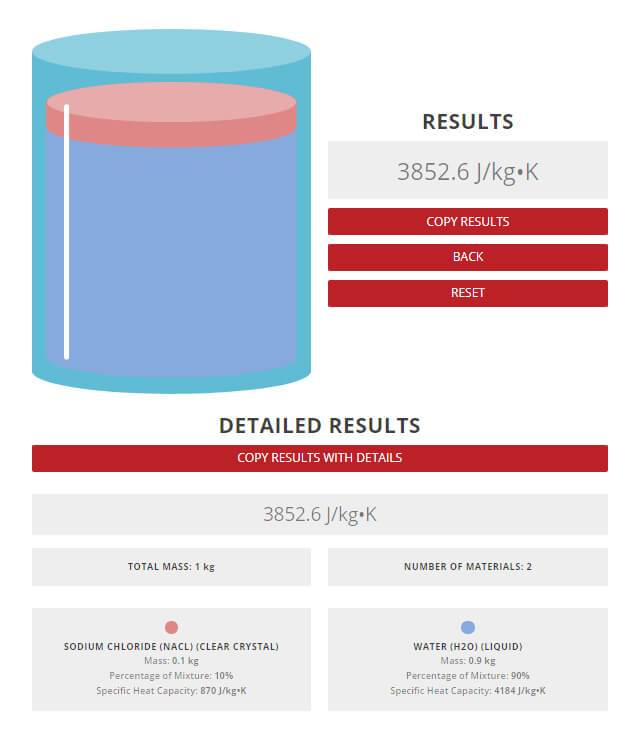
Calculate the average molar heat capacity at constant volume of a mixture containing 2 moles of monoatomic and 3 moles of diatomic ideal gas.

Calculate average molar heat capacity at constant volume of gaseous mixture contained 2 mole of each of two ideal gases A(C(v,m)=(3)/(2)R) and B(C(v,m)=(5)/(2)R) :

Calculate average molar heat capacity at constant volume of gaseous mixture contained 2 mole of each of two ideal gases A(C(v,m)=(3)/(2)R) and B(C(v,m)=(5)/(2)R) :
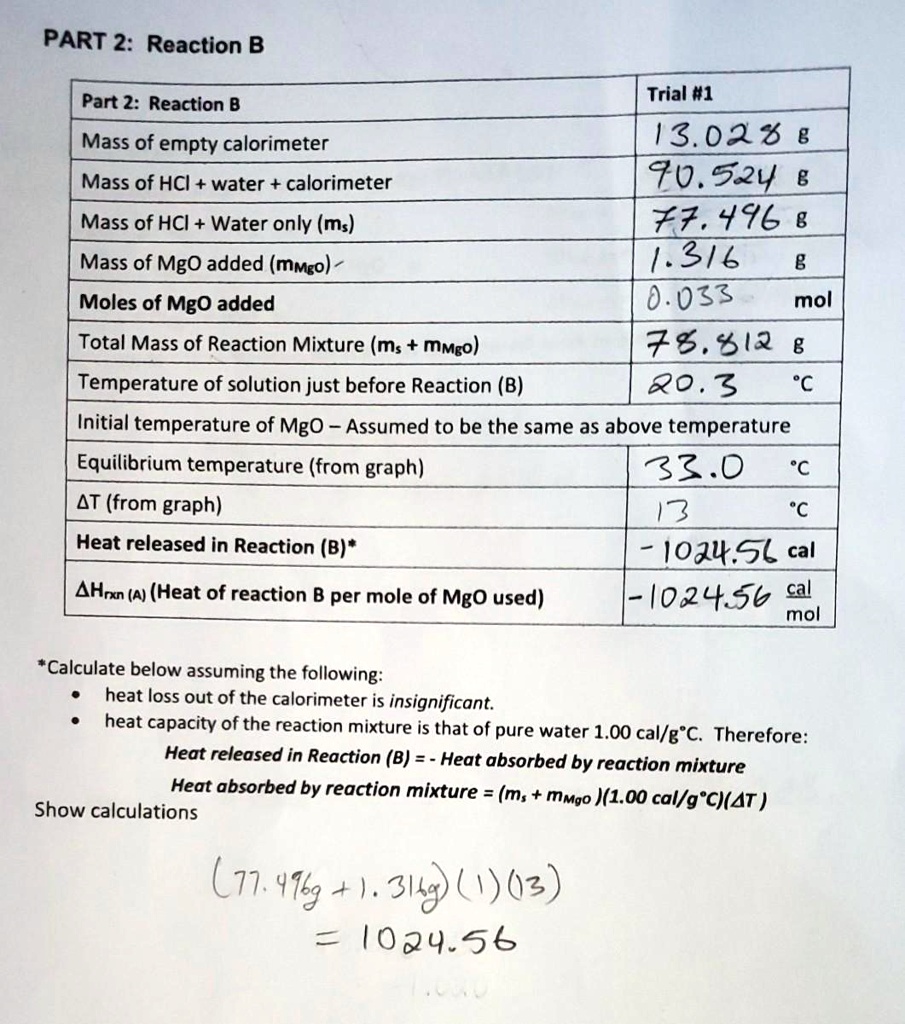
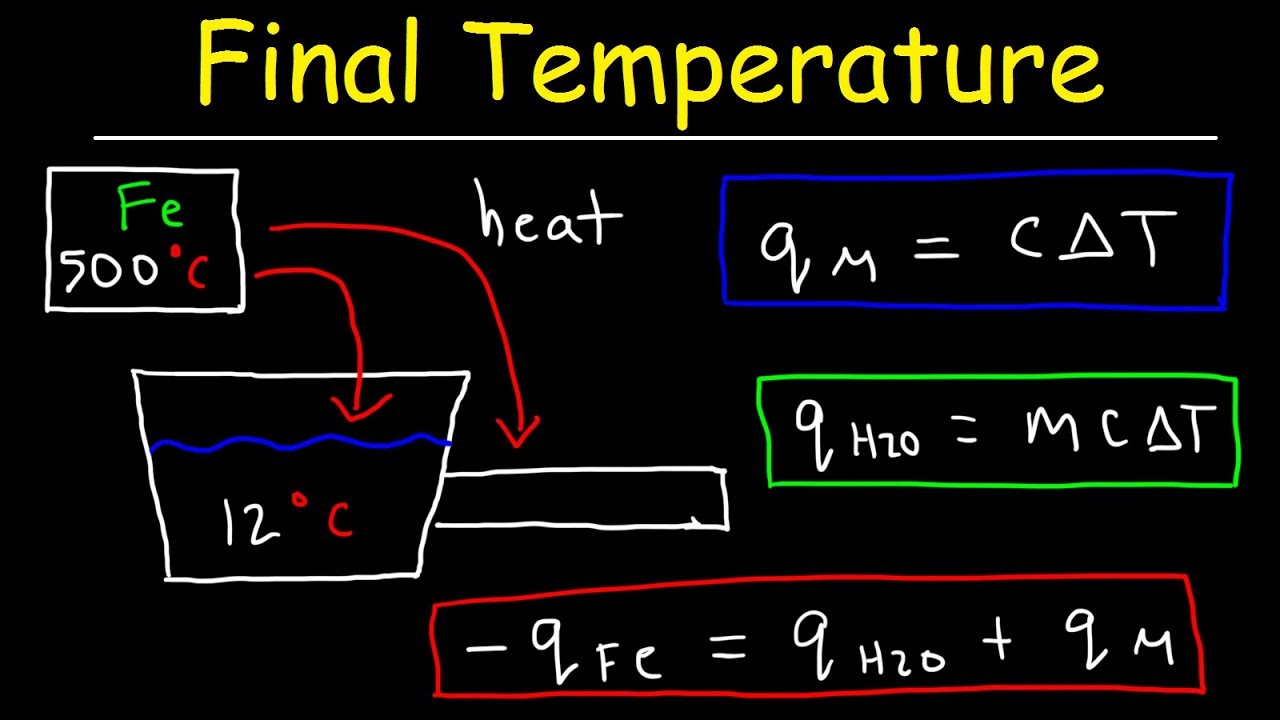
SOLVED: PART 2: Reaction B Part 2: Reaction B Mass of empty calorimeter Mass of HCI + water + calorimeter Mass of HCI Water only (ms) Trial #1 13.02 % 8 70.52y