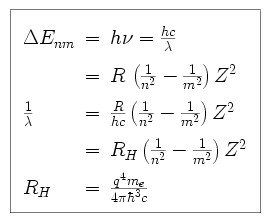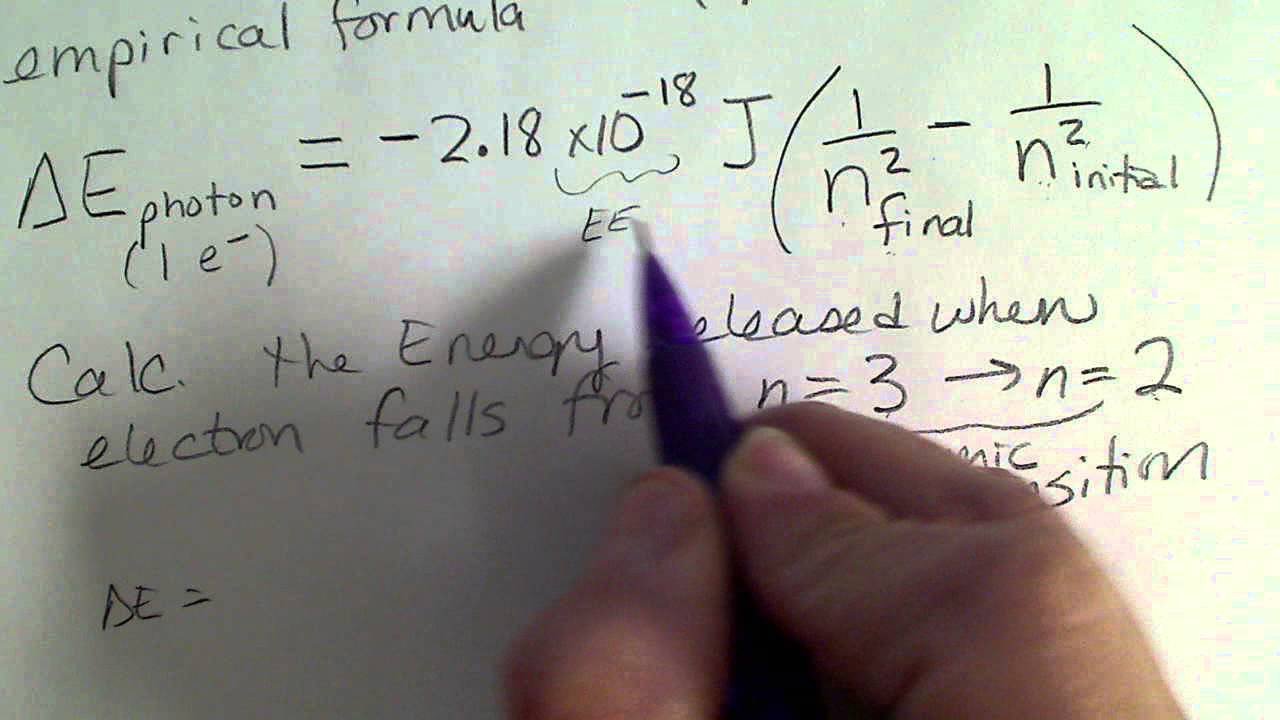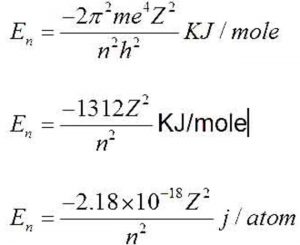Question Video: Determining the Relationship between the Atomic Radius and the Ionization Energy | Nagwa
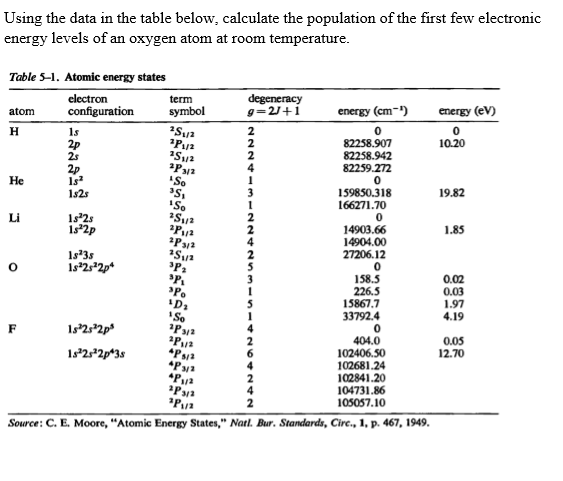
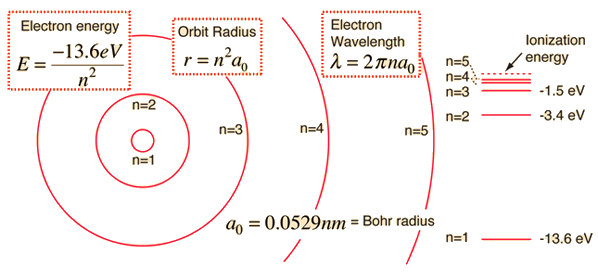
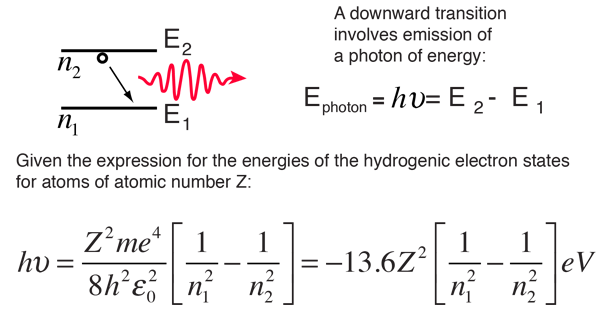
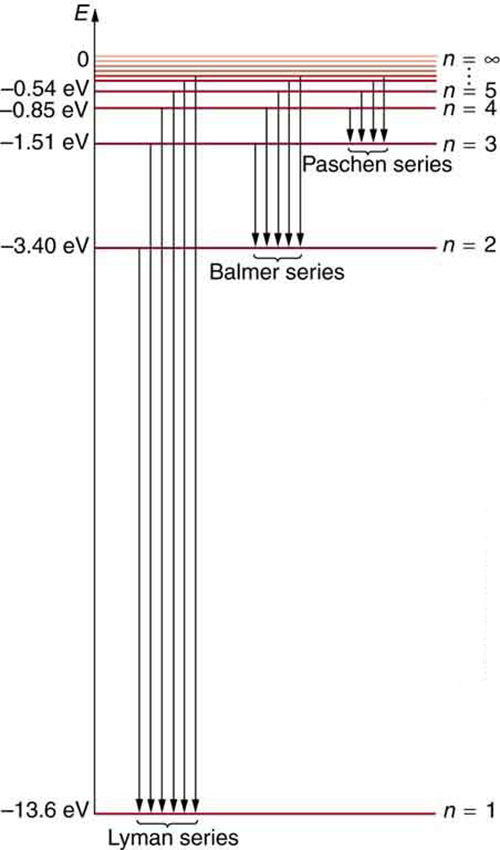
Using Bohr's formula for energy quantization, the ionisation potential of first excited state of hydrogen atom is: . (1) 13.6V, (2) 3.4V, (3) 2.6V, (4) 1.51V
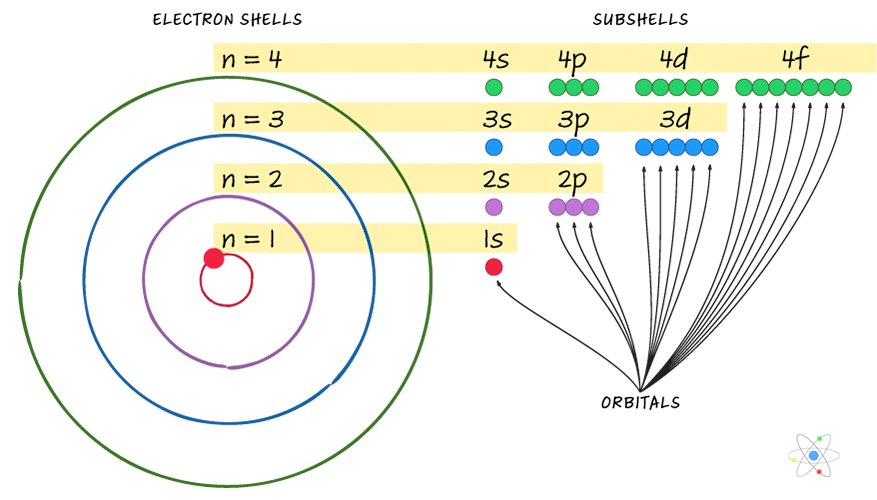
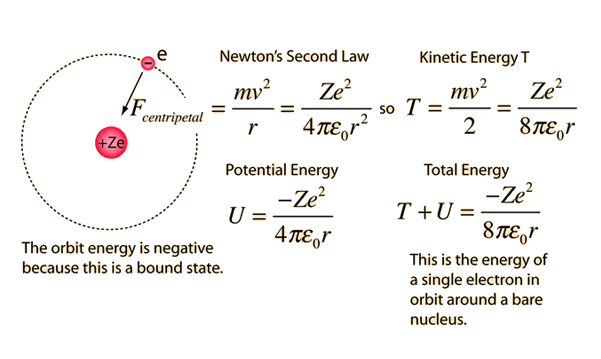
What is the formula to calculate the radius of an orbit of the atom and velocity of the specific shell of the atom.