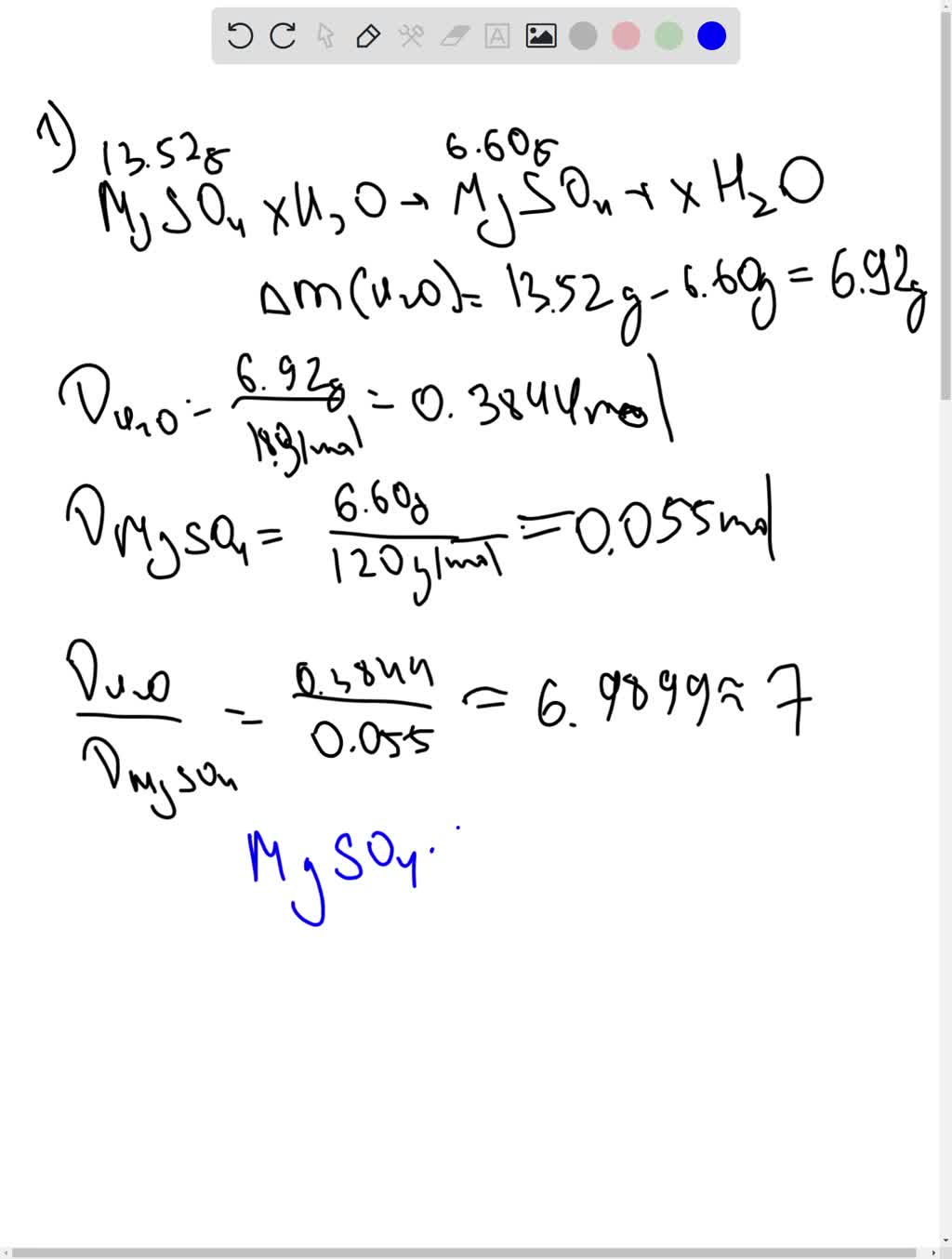Question Video: Calculating the Percentage of Water of Crystallization Given the Mass of the Hydrated and Dehydrated Salt | Nagwa

Hydrates & Anhydrates Overview, Formula & Examples | What Is an Anhydrate? - Video & Lesson Transcript | Study.com

Sample lab report - Experiment # Kayla Morren Chemical Formula of a Hydrate Objective: The objective - Studocu

SOLVED: The Mole Concept: Chemical Formula of a Hydrate Lab Report Exercise 1: Water of Hydration Data Table 1. Alum Data. Object Aluminum Cup (Empty) Aluminum Cup 2.00 grams of Alum Aluminum

Question Video: Determining the Empirical Formula of a Hydrated Salt Given the Mass of the Sample before and after Heating | Nagwa

Hydrates & Anhydrates Overview, Formula & Examples | What Is an Anhydrate? - Video & Lesson Transcript | Study.com