Evaluation of the Gibbs Free Energy Changes and Melting Temperatures of DNA/DNA Duplexes Using Hybridization Enthalpy Calculated by Molecular Dynamics Simulation | The Journal of Physical Chemistry B

Energies | Free Full-Text | Insight into the Thermodynamic Properties of Promising Energetic HNTO·AN Co-Crystal: Heat Capacity, Combustion Energy, and Formation Enthalpy

Enthalpy changes (ΔH in J g −1 ) associated with fusion (melting) of... | Download Scientific Diagram
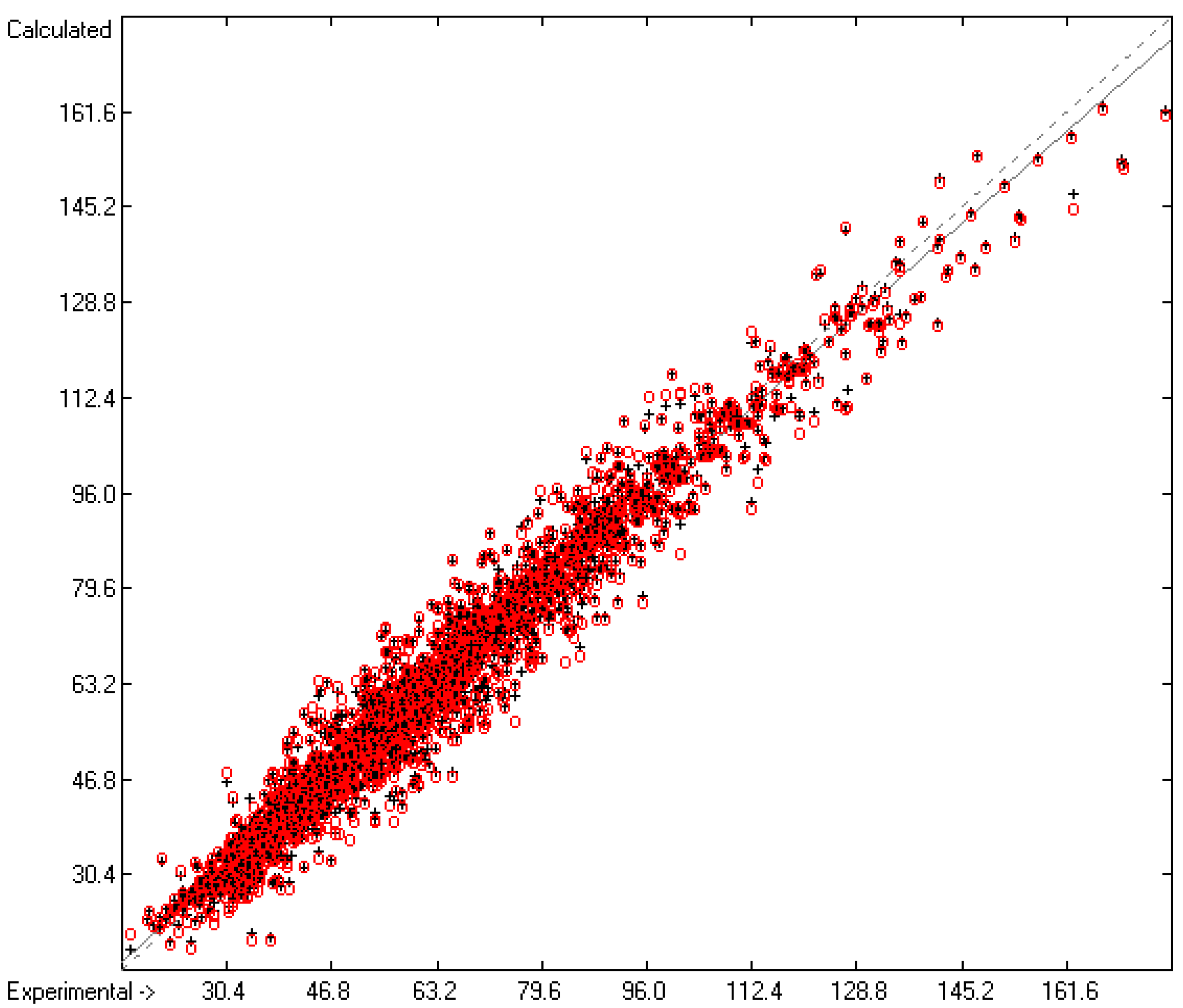
Molecules | Free Full-Text | Calculation of Five Thermodynamic Molecular Descriptors by Means of a General Computer Algorithm Based on the Group-Additivity Method: Standard Enthalpies of Vaporization, Sublimation and Solvation, and Entropy

Question Video: Calculating Standard Enthalpy of Combustion of Methane Using Standard Enthalpies of Formation of Methane and Carbon Dioxide | Nagwa
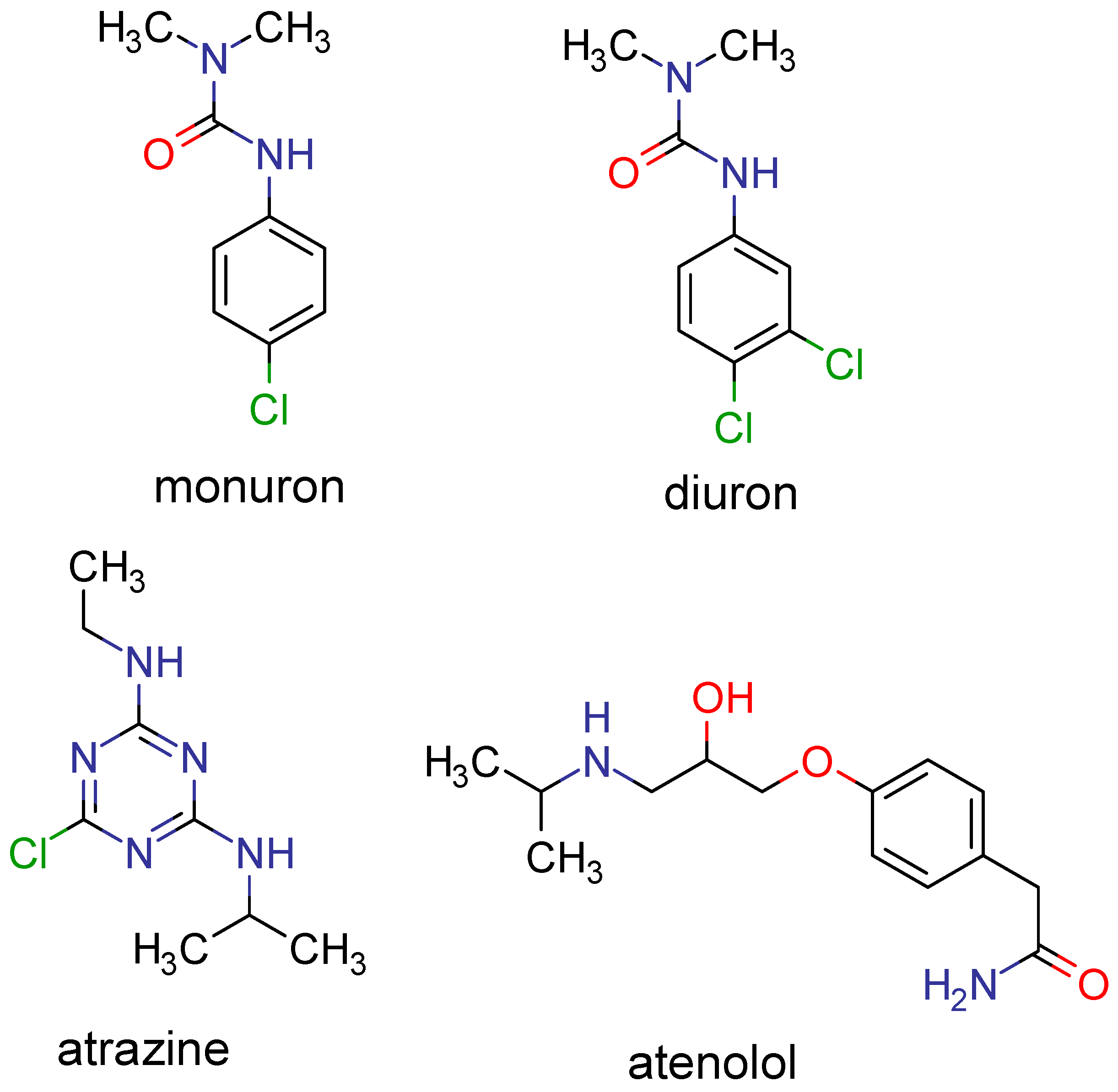
Processes | Free Full-Text | Predicting the Solubility of Nonelectrolyte Solids Using a Combination of Molecular Simulation with the Solubility Parameter Method MOSCED: Application to the Wastewater Contaminants Monuron, Diuron, Atrazine and

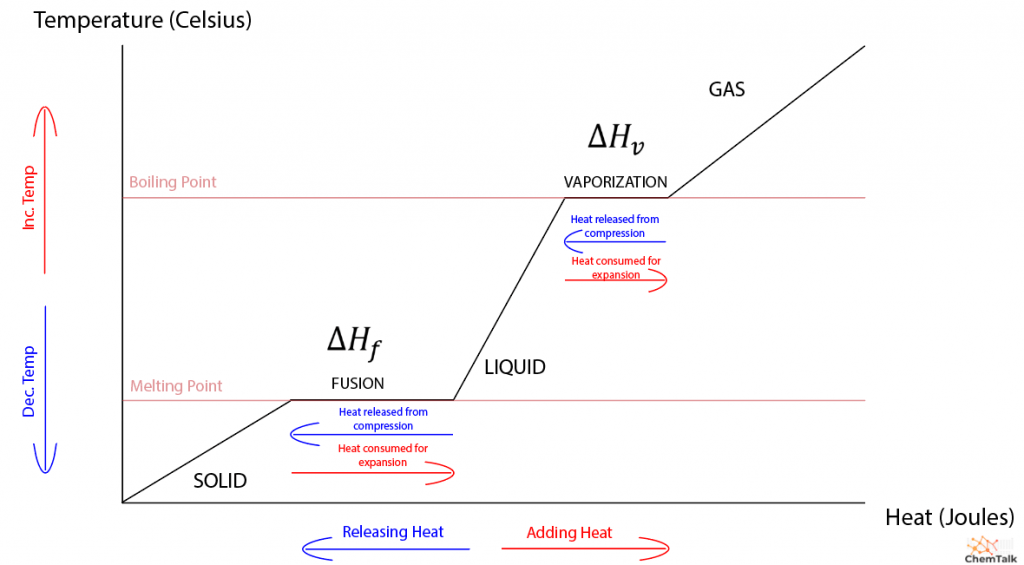
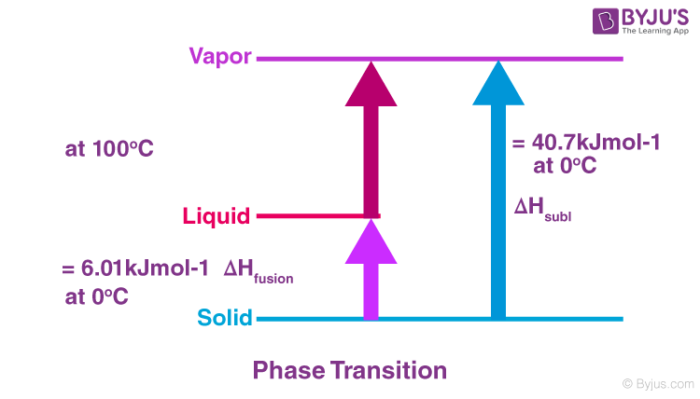
Define enthalpy of sublimation. How is it related to enthalpy of fusion and enthalpy of vaporization?
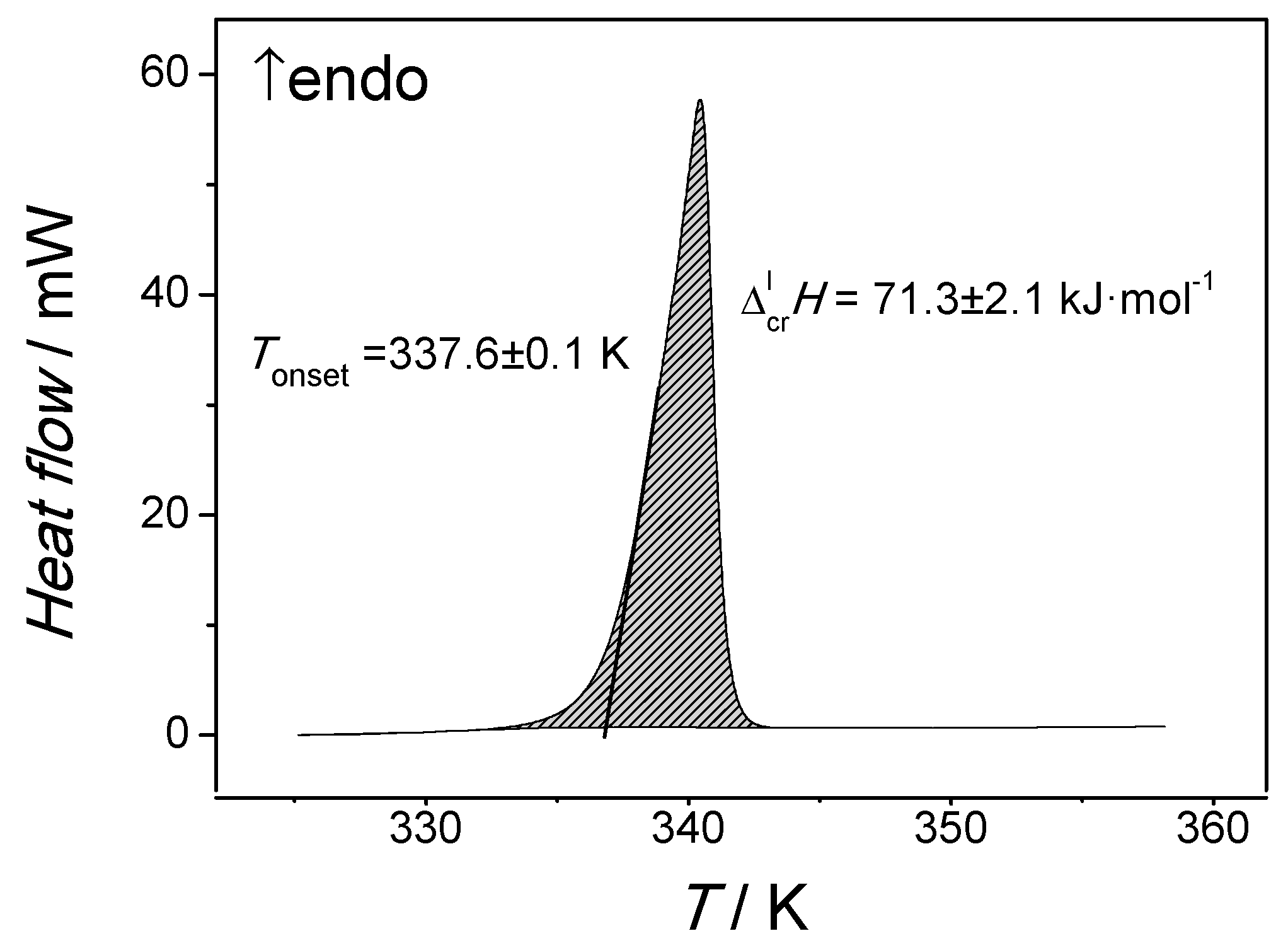
Liquids | Free Full-Text | Application of Solution Calorimetry to Determining the Fusion Enthalpy of an Arylaliphatic Compound at 298.15 K: n-Octadecanophenone

Active Thermochemical Tables: Enthalpies of Formation of Bromo- and Iodo-Methanes, Ethenes and Ethynes | The Journal of Physical Chemistry A

Latent heat of fusion of ice at 0^(@)C is 80cal/g. calculate the olar entropy change for the fusion process.

Molecular Weight Dependence of Block Copolymer Micelle Fragmentation Kinetics | Journal of the American Chemical Society