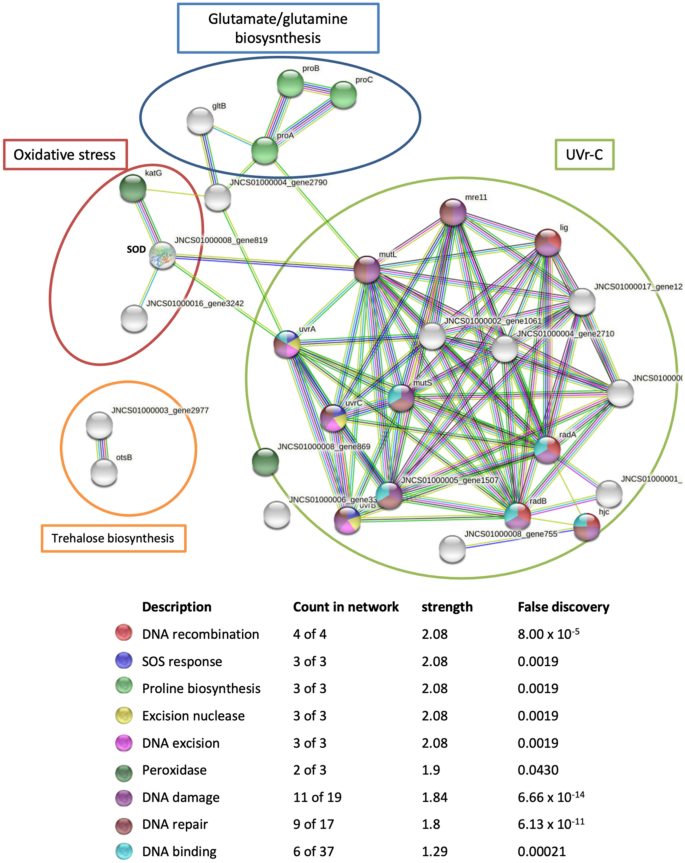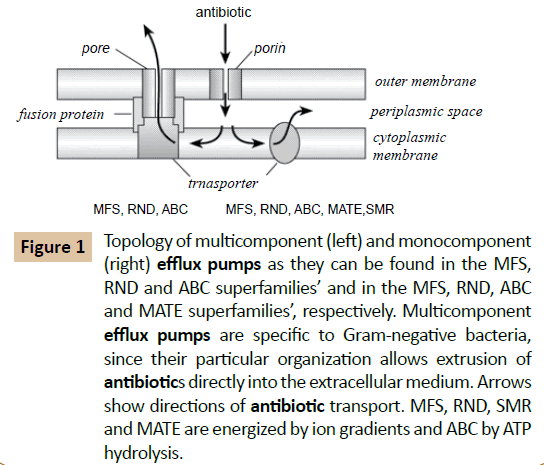
Isolation and Identification of Multi Drug Resistant Efflux Pump Protein from Klebsiella pneumoniae | Insight Medical Publishing
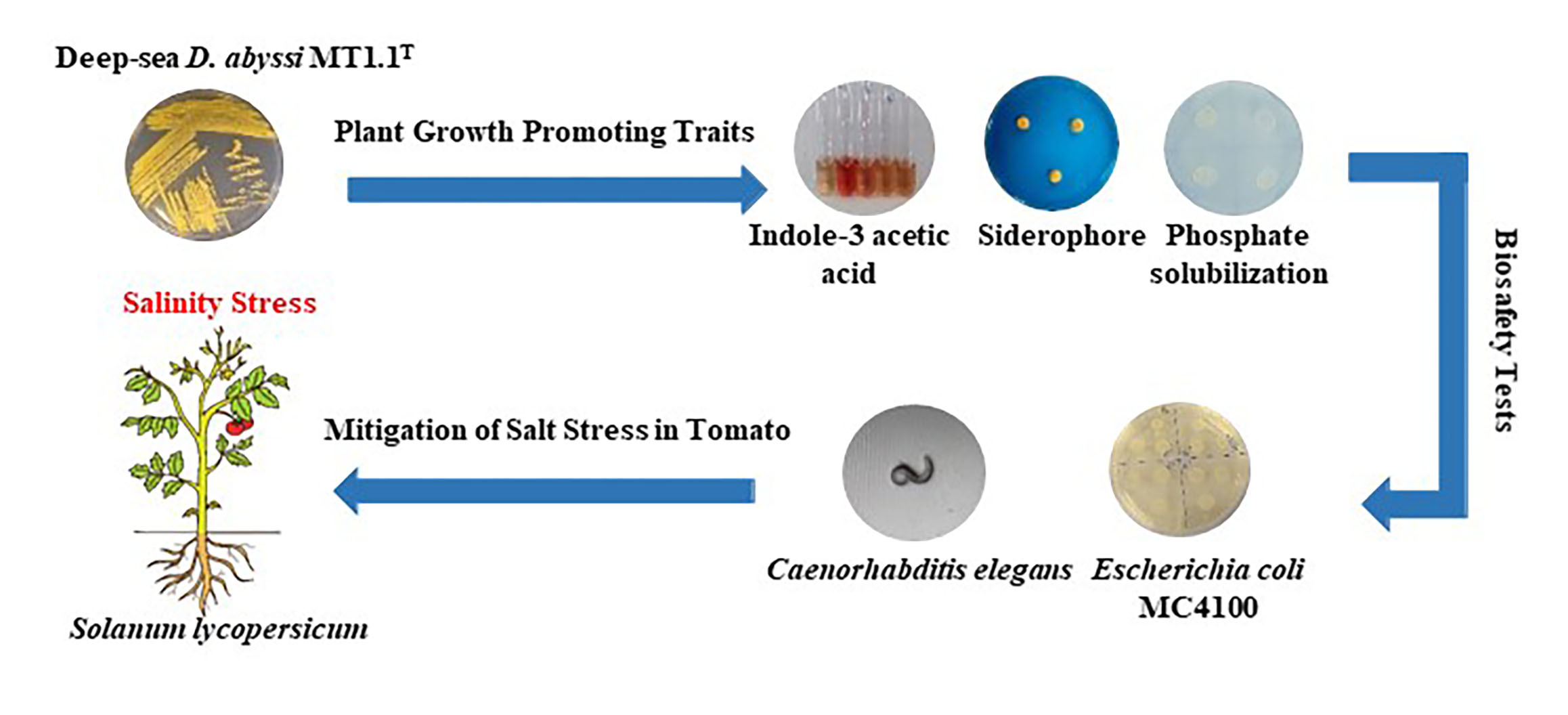
Biology | Free Full-Text | Plant Beneficial Deep-Sea Actinobacterium, Dermacoccus abyssi MT1.1T Promote Growth of Tomato (Solanum lycopersicum) under Salinity Stress

Gating of the Large Mechanosensitive Channel In Situ: Estimation of the Spatial Scale of the Transition from Channel Population Responses: Biophysical Journal

The KupA and KupB Proteins of Lactococcus lactis IL1403 Are Novel c-di-AMP Receptor Proteins Responsible for Potassium Uptake. - Abstract - Europe PMC
Global Gene Expression Analysis of Long-Term Stationary Phase Effects in E. coli K12 MG1655 | PLOS ONE

The KupA and KupB Proteins of Lactococcus lactis IL1403 Are Novel c-di-AMP Receptor Proteins Responsible for Potassium Uptake | Journal of Bacteriology

Understanding the Structural Requirements for Activators of the Kef Bacterial Potassium Efflux System | Biochemistry

The TWIK2 Potassium Efflux Channel in Macrophages Mediates NLRP3 Inflammasome-Induced Inflammation - ScienceDirect

The five classes of efflux pumps in bacteria, (i) ATP-binding cassette... | Download Scientific Diagram

A c-di-AMP riboswitch controlling kdpFABC operon transcription regulates the potassium transporter system in Bacillus thuringiensis | Communications Biology

Understanding the Structural Requirements for Activators of the Kef Bacterial Potassium Efflux System | Biochemistry
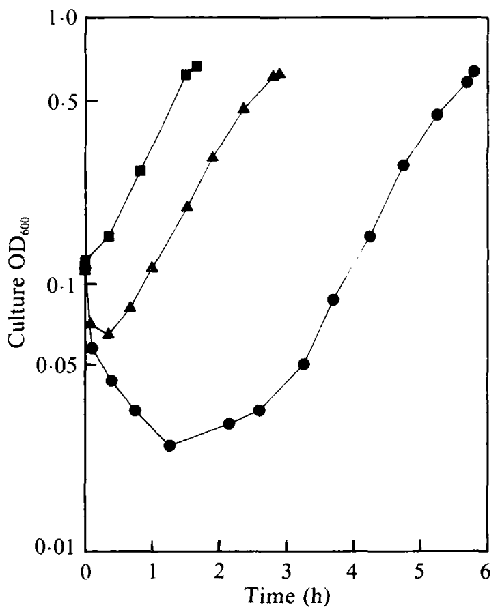
Roles of K+ and Na+ in pH homeostasis and growth of the marine bacterium Vibrio alginolyticus | Microbiology Society